Metal-binding hydrazone photoswitches for visible light reactivity and variable relaxation kinetics†
Abstract
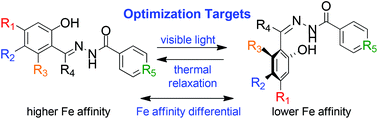
The range of applications for photoswitching moieties is diverse, and the ability to design switches with variable photochemical and physical properties is consequently important for realizing their potential. Previously we reported on the photochromism of (E)-N′-(1-(2-hydroxyphenyl)ethylidene)isonicotinohydrazide (HAPI), an aroylhydrazone compound first developed as a transition metal chelator. Herein we report the synthesis of structurally related aroylhydrazone chelators and explore the effect of these modifications on their UVA, UVC and blue light photoreactivity, photostationary state composition, photoisomer thermal stability, and relative iron(III) binding affinity. These findings will inform the next generation of aroylhydrazone photoswitches for metal-gated photoswitching applications.


 Please wait while we load your content...
Please wait while we load your content...