Advanced microscopy and spectroscopy reveal the adsorption and clustering of Cu(ii) onto TEMPO-oxidized cellulose nanofibers†
Abstract
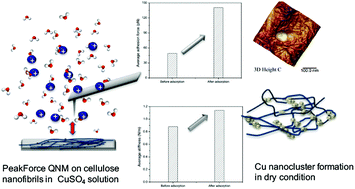
TEMPO (2,2,6,6-tetramethylpiperidine-1-oxylradical)-mediated oxidation nanofibers (TOCNF), as a biocompatible and bioactive material, have opened up a new application of nanocellulose for the removal of water contaminants. This development demands extremely sensitive and accurate methods to understand the surface interactions between water pollutants and TOCNF. In this report, we investigated the adsorption of metal ions on TOCNF surfaces using experimental techniques atthe nano and molecular scales with Cu(II) as the target pollutant in both aqueous and dry forms. Imaging with in situ atomic force microscopy (AFM), together with a study of the physiochemical properties of TOCNF caused by adsorption with Cu(II) in liquid, were conducted using the PeakForce Quantitative NanoMechanics (PF-QNM) mode at the nano scale. The average adhesion force between the tip and the target single TOCNF almost tripled after adsorption with Cu(II) from 50 pN to 140 pN. The stiffness of the TOCNF was also enhanced because the Cu(II) bound to the carboxylate groups and hardened the fiber. AFM topography, scanning electron microscopy-energy dispersive X-ray spectroscopy (SEM-EDS) mapping and X-ray photoelectron spectroscopy (XPS) indicated that the TOCNF were covered by copper nanolayers and/or nanoparticles after adsorption. The changes in the molecular structure caused by the adsorption were demonstrated by Raman and attenuated total reflectance-Fourier transform infrared spectroscopy (ATR-FTIR). This methodology will be of great assistance to gain qualitative and quantitative information on the adsorption process and interaction between charged entities in aqueous medium.


 Please wait while we load your content...
Please wait while we load your content...