Solvent-controlled reversible switching between adsorbed self-assembled nanoribbons and nanotubes†
Abstract
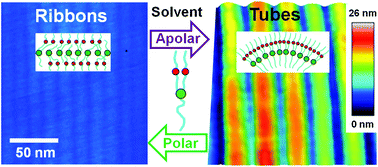
We have demonstrated that solutions of 3,5-bis-(5-hexylcarbamoylpentyloxy)-benzoic acid decyl ester (BHPB-10) can form metastable nanostructures on solid substrates and in the bulk. BHPB-10 is an achiral molecule involving several distinct, strongly interacting groups (SIGs), one aromatic-ester ring and two amide groups per molecule. Specific solvents affect the interactions between particular SIGs, thus promoting various nano-structures: lamellae, nanoribbons, helical ribbons, or nanotubes. In cyclohexane, a solvent allowing for both inter-amide hydrogen bonds and mutual attraction of rings, the formation of nanotubes with a diameter of 28 ± 5 nm was observed in the bulk and on surfaces. By contrast, in cyclohexanone, which suppresses inter-amide hydrogen bonds, flat nanoribbons with a specific width of 12 ± 4 nm were formed on solid substrates after drying. By annealing in cyclohexane vapor, we followed the process of switching surface structures from nanoribbons to nanotubes and observed helical ribbons as the precursor of nanotubes. We also turned nanotubes back into nanoribbons by adding cyclohexanone, thus demonstrating reversible switching along the route: tubes → lamellae → flat ribbons → helical ribbons → tubes. We propose models explaining the observed nanostructures and their transformations, including the origin of spontaneous chirality of the helical ribbons. Our findings on the self-assembly in the achiral BHPB-10 solutions provide insight into the influence of complementary inter-molecular specific SIG-based interactions and demonstrate an effective route for tailoring the shape and size of nanostructures derived from the same building unit.


 Please wait while we load your content...
Please wait while we load your content...