Chirality inversion, supramolecular hydrogelation and lectin binding of two thiolactose amphiphiles constructed on a di-lauroyl-l-tartaric acid scaffold†
Abstract
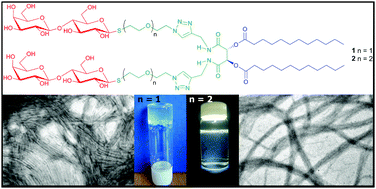
Herein we report the synthesis, characterization and self-assembly properties of two new thiolactose based amphiphiles constructed on a di-lauroyl-L-tartaric acid scaffold that only differ in the length of the spacer by an ethylene glycol unit. Upon dissolution in hot water the amphiphiles give rise to different colloidal systems at 25 °C: the one with the shorter linker forms a supramolecular thermoreversible hydrogel at a concentration of 0.1 w/v% while the other renders a colloidal system at high dilution (0.005 w/v%). Dynamic Light Scattering, Electron Microscopy (TEM, SEM and E-SEM), fluorescence CMC determination, SAXS and Circular Dichroism experiments were used to characterize both systems. The experiments indicate that only the amphiphile carrying the shorter linker is able to form a crossed-linked network of micellar fibers and thus, a stable hydrogel is observed. The difference of an ethylene glycol unit in the spacer also causes the adoption of a different molecular assembly evidenced by the inversion of the self-assembled chiral arrangement. In addition, the amphiphiles were evaluated for their ability to bind to the PNA lectin using a turbidimetric method. Agglutination was observed in both cases, a process that was disrupted upon the addition of an excess of the disaccharide lactose.


 Please wait while we load your content...
Please wait while we load your content...