Ferrocene–cinchona hybrids with triazolyl-chalcone linkers act as pro-oxidants and sensitize human cancer cell lines to paclitaxel
Abstract
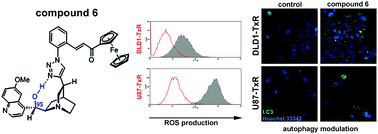
Recently, we demonstrated that ferrocene-containing compounds with a cinchona moiety displayed marked anticancer activity. Here we report on the effects of the most promising isomers encompassing quinine- (compounds 4 and 5) and quinidine-epimers (compounds 6 and 7) – synthesized using improved methods providing controlled diastereoselectivity – in three different human multidrug resistant (MDR) cancer cell lines and their sensitive counterparts (non-small cell lung carcinoma NCI-H460/R/NCI-H460, colorectal carcinoma DLD1-TxR/DLD1 and glioblastoma U87-TxR/U87). We observed that the presence of the MDR phenotype did not diminish the activity of the compounds suggesting that ferrocene quinine- and quinidine-epimers are not substrates for P-glycoprotein, which has been indicated as a major mechanism of MDR in the cell lines used. Considering that metal-based anticancer agents mainly act by increasing ROS production, we investigated the potential of ferrocene–quinidine epimers to generate ROS. We found that 6 and 7 more readily increased ROS production and induced mitochondrial damage in MDR cancer cells. According to cell death analysis, 6 and 7 were more active against MDR cancer cells showing collateral sensitivity. In addition, our data suggest that these compounds could act as inhibitors of autophagy. Importantly, simultaneous treatments of 6 and 7 with paclitaxel (PTX) increased the sensitivity of MDR cancer cells to PTX. In conclusion, the ferrocene–quinidine epimers, besides being selective towards MDR cancer cells, could also possess potential to overcome PTX resistance.

 Please wait while we load your content...
Please wait while we load your content...