Chemically coded time-programmed self-assembly†
Abstract
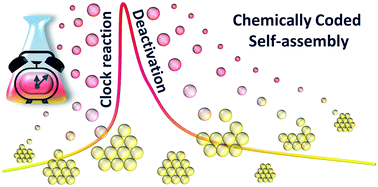
Dynamic self-assembly is of great interest in the fields of chemistry, physics and materials science and provides a flexible bottom-up approach to build assemblies at multiscale levels. We propose a method to control the time domain of self-assembling systems in a closed system, from molecular to material level using a driving chemical system: methylene glycol–sulfite pH clock reaction coupled to lactone hydrolysis. The time domain of the transient pH state (alkaline) and the time lag between the initialization of the reaction and the pH change can be efficiently fine-tuned by the initial concentration of the reagents and by the chemical composition of the lactone. The self-assembly of pH-responsive building blocks can be dynamically driven by this kinetic system, in which the time course of the pH change is coded in the system. This approach provides a flexible and autonomous way to control the self-assembly of pH responsive building blocks in closed chemical systems far from their thermodynamic equilibrium.


 Please wait while we load your content...
Please wait while we load your content...