Restoring calcium homeostasis in diabetic cardiomyocytes: an investigation through mathematical modelling
Abstract
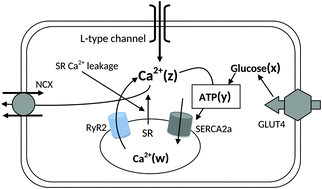
Calcium homeostasis is a key factor in the regulation of cardiac excitation–contraction coupling. Calcium dynamics in cardiomyocytes is governed by ATP which depends on insulin dependent glucose concentration, via the glucose transporter type 4 (GLUT4) transporter. It would therefore be interesting to see how calcium dynamics changes in a cardiomyocyte under diabetic conditions. We proposed and analysed a four dimensional ordinary differential equation (ODE) model to capture the interdependency of calcium dynamics on glucose uptake and ATP generation. More specifically, we looked for the role of GLUT4, energy metabolism, L-type channels, RyR2 channels, SERCA2a pumps and leakage rate in the normal functioning of cardiomyocytes. To understand the system dynamics, we first obtained the stability and Hopf-bifurcation criteria of steady state and then through parameter perturbation we captured the role of different parameters in maintaining normal calcium oscillation (frequency 40 to 180 beats per minute and amplitude ≥0.4 μM) and hence normal cardiac function. We observed that any divergence in the GLUT4 activity (especially a decrease in the glucose uptake rate) might cause abnormal calcium oscillation, leading to cardiac dysfunction (CD). Our study finally hypothesizes that a regulated sarcoplasmic reticulum (SR) calcium flux could be a possible therapeutic strategy to maintain normal calcium dynamics in diabetic heart and to prevent possible CD.


 Please wait while we load your content...
Please wait while we load your content...