Biophysical isolation and identification of circulating tumor cells†
Abstract
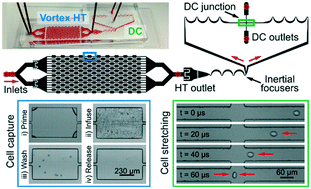
Isolation and enumeration of circulating tumor cells (CTCs) from blood is important for determining patient prognosis and monitoring treatment. Methods based on affinity to cell surface markers have been applied to both purify (via immunoseparation) and identify (via immunofluorescence) CTCs. However, variability of cell biomarker expression associated with tumor heterogeneity and evolution and cross-reactivity of antibody probes have long complicated CTC enrichment and immunostaining. Here, we report a truly label-free high-throughput microfluidic approach to isolate, enumerate, and characterize the biophysical properties of CTCs using an integrated microfluidic device. Vortex-mediated deformability cytometry (VDC) consists of an initial vortex region which enriches large CTCs, followed by release into a downstream hydrodynamic stretching region which deforms the cells. Visualization and quantification of cell deformation with a high-speed camera revealed populations of large (>15 μm diameter) and deformable (aspect ratio >1.2) CTCs from 16 stage IV lung cancer samples, that are clearly distinguished by increased deformability compared to contaminating blood cells and rare large cells isolated from healthy patients. The VDC technology demonstrated a comparable positive detection rate of putative CTCs above healthy baseline (93.8%) with respect to standard immunofluorescence (71.4%). Automation allows full enumeration of CTCs from a 10 mL vial of blood within <1 h after sample acquisition, compared with 4+ hours with standard approaches. Moreover, cells are released into any collection vessel for further downstream analysis. VDC shows potential for accurate CTC enumeration without labels and confirms the unique highly deformable biophysical properties of large CTCs circulating in blood.
- This article is part of the themed collection: Lab on a Chip Pioneers of Miniaturization Lectureship winners


 Please wait while we load your content...
Please wait while we load your content...