Separation and dual detection of prostate cancer cells and protein biomarkers using a microchip device†
Abstract
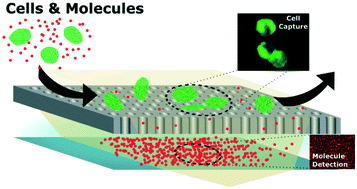
Current efforts for the detection of prostate cancer using only prostate specific antigen are not ideal and indicate a need to develop new assays – using multiple targets – that can more accurately stratify disease states. We previously introduced a device capable of the concurrent detection of cellular and molecular markers from a single sample fluid. Here, an improved design, which achieves affinity as well as size-based separation of captured targets using antibody-conjugated magnetic beads and a silicon chip containing micro-apertures, is presented. Upon injection of the sample, the integration of magnetic attraction with the micro-aperture chip permits larger cell–bead complexes to be isolated in an upper chamber with the smaller protein–bead complexes and remaining beads passing through the micro-apertures into the lower chamber. This enhances captured cell purity for on chip quantification, allows the separate retrieval of captured cells and proteins for downstream analysis, and enables higher bead concentrations for improved multiplexed ligand targeting. Using LNCaP cells and prostate specific membrane antigen (PSMA) to model prostate cancer, the device was able to detect 34 pM of spiked PSMA and achieve a cell capture efficiency of 93% from culture media. LNCaP cells and PSMA were then spiked into diluted healthy human blood to mimic a cancer patient. The device enabled the detection of spiked PSMA (relative to endogenous PSMA) while recovering 85–90% of LNCaP cells which illustrated the potential of new assays for the diagnosis of prostate cancer.
- This article is part of the themed collection: RSC papers by NanoEngineering for Medicine and Biology 2018 Speakers


 Please wait while we load your content...
Please wait while we load your content...