Natural Fe isotope fractionation in an intestinal Caco-2 cell line model†
Abstract
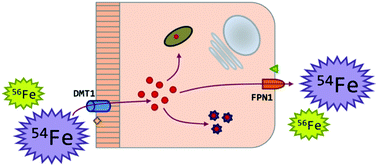
In this work, Fe isotopic analysis of samples obtained from an in vitro intestinal model was performed via multi-collector ICP-mass spectrometry (MC-ICP-MS) to evaluate the isotope fractionation accompanying Fe uptake and transport mechanisms at a cellular level. The Caco-2 cell line has been used, after cell differentiation, as an enterocyte model and a bi-cameral experimental setup has been developed and optimized for stimulating intracellular Fe fluxes. An Fe : ascorbic acid mixture with a molar ratio of 1 : 5 was used as a source of non-heme bioavailable Fe. Good experimental repeatability and reproducibility were attained with low blank contribution levels, allowing precise and reliable Fe isotope ratio results. Both Fe absorption and transport processes were accompanied by Fe isotope fractionation in favor of the lighter isotopes. After 3 hours of exposure, the isotopic composition of the apical solution and the cells did not significantly differ from that of the original solution added to the cells. After 24 hours of exposure, the trend observed was towards a light Fe isotopic composition in the cells, whereas the apical solutions were enriched in the heavier isotopes. These results were in good agreement with previous in vivo and ex vivo findings. An overall increase in delta Fe values of the cell layers exposed to Fe treatment relative to the corresponding values for the untreated cells also seems to support the assumption of a preferential accumulation of heavy isotopes in enterocyte ferritin. The consistency of the results obtained supports the usefulness of in vitro cell culture models as an interesting complementary tool for studying Fe metabolic pathways at the intestinal level.
- This article is part of the themed collections: JAAS Emerging Investigator Lectureship winners, JAAS Recent HOT articles and Young Analytical Scientists


 Please wait while we load your content...
Please wait while we load your content...