Catalytic fast pyrolysis of biomass: superior selectivity of hierarchical zeolites to aromatics†
Abstract
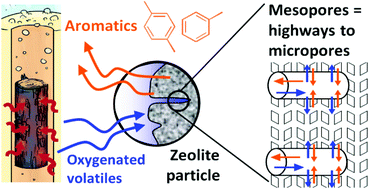
The catalytic fast pyrolysis of oak over two zeolites (microporous and hierarchical) was investigated in a microfluidized bed reactor (MFBR) at 500 °C and as a function of the biomass-to-catalyst ratio. A hierarchical zeolite was produced by desilication with a NaOH solution of a conventional HZSM-5 zeolite. The outlet of the MFBR was connected to a single photoionization mass spectrometer (SPI-MS) for the on-line analysis of volatiles. This on-line analysis method allows studying the dynamics of volatile formation (in real time) and the deactivation of 2 zeolites upon stepwise injections of wood particles. Strikingly, the selectivity of targeted mono-aromatic compounds (quantified by gas chromatography) is doubled after desilication of the zeolite. The coked zeolites were characterized by TEM-EDX, digestion in fluoric acid, MALDI-TOF MS, etc. Three different types of coke are evidenced: (1) coke trapped inside micropores, (2) external coke formed on the outer surface of the crystals and (3) coke precursors deposited in the mesopores. The latter two cokes are much less toxic than the microporous coke. The open mesopores produced after desilication can be seen as “highways” where big molecules (such as levoglucosan) can diffuse to more accessible pore mouths. Brønsted acid sites present on microporous mouths can be active for the conversion of bigger molecules to small fragments. These fragments diffuse and form aromatics in the micropores (shape selectivity). The mesopores also promote the evacuation of catalytic products, thus enhancing the selectivity of mono-aromatic hydrocarbons. Desilicated zeolites present higher selectivity to mono-aromatics and stability upon coke deposition than microporous zeolites.


 Please wait while we load your content...
Please wait while we load your content...