Production of levoglucosenone and 5-hydroxymethylfurfural from cellulose in polar aprotic solvent–water mixtures†
Abstract
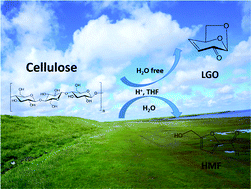
We demonstrate a process to produce levoglucosenone (LGO) and 5-hydroxymethylfurfural (HMF) from cellulose in up to 65% carbon yield using sulfuric acid as catalyst and a solvent consisting of a mixture of tetrahydrofuran (THF) with water. In pure THF, LGO is the major product of cellulose dehydration, passing through levoglucosan as an intermediate. Increasing the water content (up to 5 wt%) results in HMF as the major product. HMF is formed both by glucose dehydration and direct dehydration of LGA. The maximum combined yield of LGO and HMF (∼65 carbon%) is achieved in the presence of 1–2.5 wt% H2O, such that comparable amounts of these two co-products are formed. THF gave the highest total yields of LGO and HMF among the solvents investigated in this study (i.e., THF, diglyme, tetraglyme, gamma-valerolactone (GVL), cyclopentyl methyl ether (CPME), 1,4-dioxane, and dimethyl sulfoxide (DMSO)). Furthermore, the rate of LGO and HMF degradation in THF was lower than in the other solvents. LGO/HMF yields increased with increased strength of the acid catalyst (H2SO4 > H3PO4 > HCOOH), and HMF was produced more selectively than LGO in the presence of hydrochloric acid. Techno-economic analysis for LGO and HMF production from cellulose shows that the lowest LGO/HMF production costs are less than $3.00 per kg and occur at a cellulose loading and water content of 2–3% and 1.5–2.5% respectively.


 Please wait while we load your content...
Please wait while we load your content...