Design of a Cu(i)/C-doped boron nitride electrocatalyst for efficient conversion of CO2 into acetic acid†
Abstract
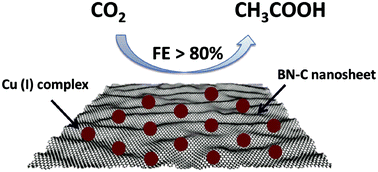
Electrocatalytic CO2 reduction to C2 products is a promising technique when compared with the traditionally complicated and energy intensive routes in the industrial process. As an important bulk chemical, it is highly desirable for acetic acid to be produced via a sustainable method. In this work, we prepared N-based Cu(I)/C-doped boron nitride (BN-C) composites for electrocatalytic reduction of CO2 to acetic acid. It was found that the Faradaic efficiency of acetic acid could reach as high as 80.3% with a current density of 13.9 mA cm−2 when 1-ethyl-3-methylimidazolium tetrafluoroborate ([Emim]BF4)-LiI-water solution was used as the electrolyte, which was about 4 times higher than the best value reported in the literature. Detailed studies further indicated that the Cu complex, BN-C, and the electrolyte have an excellent synergistic effect for producing acetic acid. In particular, as a promoter, LiI played a key role in C–C coupling to form acetic acid in the electrocatalytic process. Our study shows a promising way to produce C2 products via electrochemical reduction of CO2 by the combination of composite electrodes and electrolytes with a promoter.


 Please wait while we load your content...
Please wait while we load your content...