Preparation of optically active cycloalkenes bearing all-carbon quaternary stereogenic centres via lipase–oxovanadium combo-catalysed dynamic kinetic resolution†
Abstract
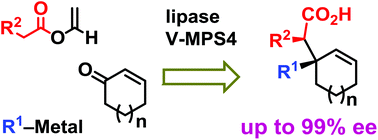
In this study, a novel asymmetric synthesis of all-carbon quaternary stereogenic centres is developed by the connection of three prochiral or achiral components—conjugated enones, organometallic compounds and vinyl esters—at the C-1 position of the enones. This method involves three sequential steps: 1,2-nucleophilic addition of an organometallic compound to the enone, lipase-catalysed dynamic kinetic resolution (DKR) of the tert-allylic alcohol and the Ireland–Claisen rearrangement of the optically active allyl ester thus generated. This method features the effective use of acyl moieties installed by DKR for achieving high atom economy. The application of this method to the protective-group-free asymmetric total synthesis of (−)-crinane, a core structure of a class of natural alkaloids, has demonstrated that it can alter a known synthetic pathway of a racemate into an asymmetric synthesis of an optically pure molecule while reducing the total transformation steps and increasing the overall yield. With these advantages, this method is practical and attractive as a new environmentally benign protocol.
- This article is part of the themed collections: 2017 Green Chemistry Hot Articles and Enzyme catalysis in organic synthesis

 Please wait while we load your content...
Please wait while we load your content...