Enhanced biofilm penetration for microbial control by polyvalent phages conjugated with magnetic colloidal nanoparticle clusters (CNCs)†
Abstract
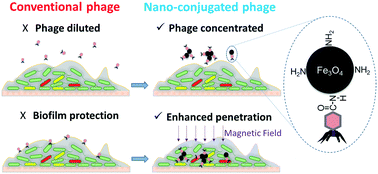
Biofilms may shelter pathogenic or other problematic microorganisms that are difficult to eradicate due to hindered penetration of antimicrobial chemicals. Here, we demonstrate the potential for efficient bacterial suppression using polyvalent (broad host-range) phages attached to magnetic colloidal nanoparticle clusters (CNCs) that facilitate biofilm penetration under a relatively small magnetic field (660 gauss). The polyvalent phage PEL1 (Podoviridae family) was immobilized onto Fe3O4-based magnetic CNCs that had been coated with chitosan (and thus functionalized with amino groups). This facilitated conjugation with phages via covalent bonding (i.e., amide linkages) and enabled phage loading, which reached (5.2 ± 0.7) × 103 centers of infection per 1 μg of chitosan-coated CNCs (CS-Fe3O4). The plaque formation capability of PEL1–CS-Fe3O4 on Pseudomonas aeruginosa PA01 and Escherichia coli C3000 lawns was significantly higher than that of phages conjugated with similar CNCs that had been functionalized with carboxyl groups (99.1% vs. 3.2% Petri dish area of infection). In newly established biofilms formed from these two species on a glass surface, PEL1–CS-Fe3O4 removed 88.7 ± 2.8% of the biofilm coverage area after 6 h of treatment. Overall, this conjugation approach could extend the application of phages for microbial control by enhancing their delivery to relatively inaccessible locations within biofilms.


 Please wait while we load your content...
Please wait while we load your content...