Polypyrrole-assisted oxygen electrocatalysis on perovskite oxides†
Abstract
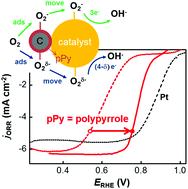
Nitrogen-containing electrocatalysts, such as metal–nitrogen–carbon (M–N–C) composites and nitrogen-doped carbons, are known to exhibit high activities for an oxygen reduction reaction (ORR). Moreover, even if the mechanism by which nitrogen improves the activities is not completely understood, a strong electronic interaction between nitrogen and active sites has been found in these composites. Herein, we demonstrate a case in which nitrogen improves the electroactivity, but in the absence of a strong interaction with other components. The overpotentials of the ORR and oxygen evolution reaction (OER) on perovskite oxide catalysts were significantly reduced simply by mixing the catalyst particles with polypyrrole/carbon composites (pPy/C). Any strong interactions between pPy (a nitrogen-containing compound) and active sites of the catalysts are not confirmed. A scenario based on the sequential task allocation between pPy and oxide catalysts for the ORR was proposed: (1) molecular oxygen is incorporated into pPy as a form of superoxide (pPy+O2−), (2) the superoxide is transferred to the active sites of perovskite catalysts, and (3) the superoxide is completely reduced along the 4e ORR process.


 Please wait while we load your content...
Please wait while we load your content...