Self-polymerized dopamine as an organic cathode for Li- and Na-ion batteries†
Abstract
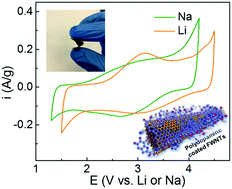
Self-polymerized dopamine is a versatile coating material that has various oxygen and nitrogen functional groups. Here, we demonstrate the redox-active properties of self-polymerized dopamine on the surface of few-walled carbon nanotubes (FWNTs), which can be used as organic cathode materials for both Li- and Na-ion batteries. We reveal the multiple redox reactions between self-polymerized dopamine and electrolyte ions in the high voltage region from 2.5 to 4.1 V vs. Li using both density functional theory (DFT) calculations and electrochemical measurements. Free-standing and flexible hybrid electrodes are assembled using a vacuum filtration method, which have a 3D porous network structure consisting of polydopamine coated FWNTs. The hybrid electrodes exhibit gravimetric capacities of ∼133 mA h g−1 in Li-cells and ∼109 mA h g−1 in Na-cells utilizing double layer capacitance from FWNTs and multiple redox-reactions from polydopamine. The polydopamine itself within the hybrid film can store high gravimetric capacities of ∼235 mA h g−1 in Li-cells and ∼213 mA h g−1 in Na-cells. In addition, the hybrid electrodes show a high rate-performance and excellent cycling stability, suggesting that self-polymerized dopamine is a promising cathode material for organic rechargeable batteries.
- This article is part of the themed collection: 2017 Energy and Environmental Science HOT articles


 Please wait while we load your content...
Please wait while we load your content...