An investigation into support cooperativity for the deoxygenation of guaiacol over nanoparticle Ni and Rh2P†
Abstract
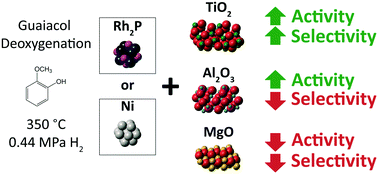
The production of hydrocarbon fuels from biomass pyrolysis requires the development of effective deoxygenation catalysts, and insight into how the properties of the support influence performance is critical for catalyst design. In this report, nanoparticles of Ni and Rh2P were synthesized using solution-phase techniques and dispersed on high surface area supports. The supports included a relatively inert material (C), an acidic reducible metal-oxide (TiO2), an acidic irreducible metal-oxide (Al2O3), and a basic irreducible metal-oxide (MgO). The eight active phase/support combinations were investigated for the deoxygenation of guaiacol, a pyrolysis vapor model compound, under ex situ catalytic fast pyrolysis conditions (350 °C, 0.44 MPa H2). Compared to the baseline performance of the C-supported catalysts, Ni/TiO2 and Rh2P/TiO2 exhibited higher guaiacol conversion and lower O : C ratios for C5+ products, highlighting the enhanced activity and greater selectivity to deoxygenated products derived from the use of an acidic reducible metal-oxide support. The Al2O3-supported catalysts also exhibited higher conversion than the C-supported catalysts and promoted alkylation reactions, which improve carbon efficiency and increase the carbon number of the C5+ products. However, Ni/Al2O3 and Rh2P/Al2O3 were less selective towards deoxygenated products than the C-supported catalysts. The MgO-supported catalyst exhibited lower conversion and decreased yield of deoxygenated products compared to the C-supported catalysts. The results reported here suggest that basic metal-oxide supports may inhibit deoxygenation of phenolics under CFP conditions. Contrastingly, support acidity and reducibility were demonstrated to promote conversion and selectivity to deoxygenated products, respectively.


 Please wait while we load your content...
Please wait while we load your content...