Unraveling the surface state and composition of highly selective nanocrystalline Ni–Cu alloy catalysts for hydrodeoxygenation of HMF†
Abstract
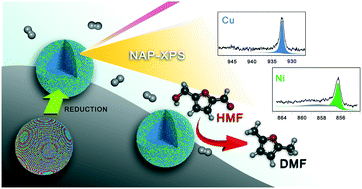
The selective hydrodeoxygenation (HDO) of 5-hydroxymethylfurfural (HMF) to 2,5-dimethylfuran (DMF) is an important step in cellulosic biomass upgrading to biofuels, where bimetallic oxophilic catalysts have shown promising performance. Well controlled bimetallic NiCu and NiCu3 nanocrystals supported on carbon are shown to give high yields and selectivities to DMF. To shed light on the active phase, near-ambient-pressure X-ray photoelectron spectroscopy (NAP-XPS) was used to characterize the surface composition of these highly selective base-metal catalysts under reducing conditions relevant to the HDO reaction. Reactions were performed in a continuous flow reactor under reasonable conditions of 33 bar and 180 °C. The Ni alloys were significantly more selective for DMF compared to monometallic Ni or Cu catalysts. With a well-controlled surface composition, the nanocrystal NiCu3/C catalyst exhibited a maximum DMF yield of 98.7%. NAP-XPS characterization showed that the Ni–Cu nanocrystals were completely reduced below 250 °C in H2; this, together with bulk thermodynamic calculations, implies that the catalysts were completely reduced under the reaction conditions. NAP-XPS also indicated that the NiCu3 nanocrystal structure consisted of a Cu-rich core and a 1 : 1 molar Ni : Cu shell.


 Please wait while we load your content...
Please wait while we load your content...