Effect of hierarchical porosity in Beta zeolites on the Beckmann rearrangement of oximes
Abstract
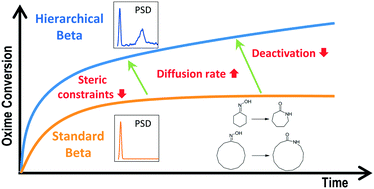
Hierarchical Beta zeolites with different Si/Al molar ratios, synthesized by crystallization of silanized protozeolitic units, were investigated in the liquid-phase Beckmann rearrangement of cyclohexanone and cyclododecanone oximes. The hierarchical Beta samples contain mesopores with sizes in the range 2.0–6.0 nm, as well as the typical zeolite micropores, showing a great contribution of the mesopore/external surface area (300–328 m2 g−1) to the BET surface area (706–763 m2 g−1) and increased Lewis acidity compared to conventional Beta zeolites. Diffusional and steric limitations were found to strongly influence the oxime Beckmann rearrangement as enhanced conversions were obtained with the hierarchical Beta zeolites. The largest differences between conventional and hierarchical Beta zeolites were denoted with the bulkier oxime, i.e. in the rearrangement of cyclododecanone oxime. In this case, two stages are clearly observed in the evolution of the oxime conversion along the reaction time, with a plateau being reached at long reaction times due to deactivation by product inhibition. Hierarchical Beta zeolites exhibited superior performance in both stages compared to reference samples. These results indicate that the use of hierarchical Beta zeolites for oxime Beckmann rearrangement leads to significant improvements in the catalytic behavior compared to conventional Beta samples due to a number of factors: faster intracrystalline diffusion, availability of non-sterically hindered mesopore/surface area and lower deactivation through product inhibition.

 Please wait while we load your content...
Please wait while we load your content...