Synthesis of sinter-resistant Au@silica catalysts derived from Au25 clusters†
Abstract
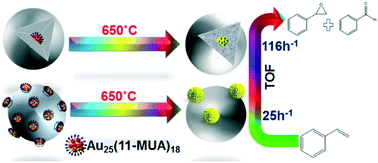
Gold clusters exhibit remarkable catalytic activity for many reactions such as carbon monoxide oxidation, alcohol, alkene, and hydrocarbon oxidations, and reduction reactions at low temperatures. However, several previous studies show that Au clusters undergo problematic sintering at temperatures above 250 °C, which makes them unsuitable catalysts for high-temperature oxidation reactions. Here we report the coating of Au25(11-MUA)18 clusters (where 11-MUA = mercaptoundecanoic acid) by silica to produce sinter-resistant Au@SiO2 catalysts. The structure of the resulting materials before and after calcination at temperatures up to 650 °C was followed by TEM and extended X-ray absorption fine structure spectroscopy (EXAFS) analyses, which showed that the Au@SiO2 catalysts created were much more stable to sintering compared to control materials; with average particles sizes of 2.2 nm after calcination at 250 °C and just over 3 nm after calcination at 650 °C. In addition, we explored the activity of the resulting materials for the 4-nitrophenol reduction and styrene epoxidation reactions; results clearly showed that the Au surfaces are accessible for reactants and that the kinetics of 4-nitrophenol reduction was directly related to the dispersion of the Au particles, as measured via the first shell Au–Au coordination numbers by EXAFS. Styrene epoxidation results show that the Au@SiO2 materials have excellent activity and recyclability.
- This article is part of the themed collection: 2017 Catalysis Science & Technology HOT Articles


 Please wait while we load your content...
Please wait while we load your content...