The duality of UiO-67-Pt MOFs: connecting treatment conditions and encapsulated Pt species by operando XAS†
Abstract
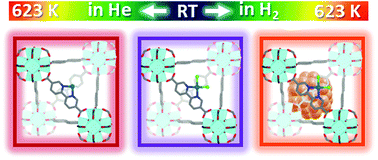
An X-ray absorption spectroscopy study of the UiO-67 Pt functionalized metal organic frameworks (MOFs) demonstrates that under appropriate conditions, at least two types of catalytically active sites can be formed in the cavities of the MOF: isolated Pt-complexes and Pt nanoparticles (Pt-NPs). Both pre-made linker synthesis (PMLS) and post-synthesis functionalization (PSF) methods were adopted. XAS was used to monitor the temperature-dependent behaviour of UiO-67-Pt while heating from RT to 623 K, in different gas feeds (pure He, 3% H2/He and 10% H2/He). We collected static in situ Pt LIII XANES and EXAFS spectra at room temperature (RT) before and after the thermal treatment, as well as spectra acquired under operando conditions upon heating. Under 10% H2/He thermal treatment, we unambiguously detected Pt-NP formation which has been followed by a parametric EXAFS analysis of the data collected during temperature programmed H2-reduction (TPR), using the Einstein model to predict the temperature dependence of the Debye–Waller factors. Conversely, in pure He flow, the only significant change observed during TPR is the progressive decrease of the Pt–Cl single scattering contribution, leading to the conclusion that the Pt grafted to the bpydc-linkers remains naked. Advanced EXAFS/TEM analysis allowed us to quantify the fraction of Pt in the form of Pt-NPs, values that have been quantitatively confirmed by linear combination analysis of the XANES spectra. In situ XANES/EXAFS study was supported by ex situ XRPD and BET analyses, confirming the framework stability and testifying a loss of the internal volume after TPR due to the formation of Pt-NPs insides the MOF pores, more relevant in the sample where smaller Pt-NPs were formed.


 Please wait while we load your content...
Please wait while we load your content...