Packing arrangement of 13C selectively labeled sequence model peptides of Samia cynthia ricini silk fibroin fibers studied by solid-state NMR†
Abstract
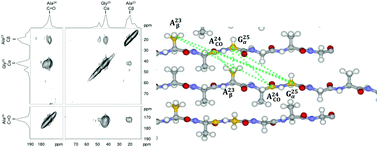
Samia cynthia ricini (S. c. ricini) is one of the wild silkworms. Their silk fibroins have been paid attention as potentially valuable biomedical materials as well as Bombyx mori silk fibroins, but detailed information on the packing arrangement of the fibers is still not currently well understood at a molecular level. In this study, 34 mer model peptides, GGAGGGYGGDGG(A)12GGAGDGYGAG with different 13C labeled positions have been synthesized as a typical sequence of the primary structure of S. c. ricini silk fibroins made up of tandemly repeated sequences of polyalanine as the crystalline region and glycin-rich sequences as the non-crystalline region. The heterogeneous structure was obtained from the determination of the fraction of several conformations depending on the position of the Ala residue by 13C cross polarization/magic angle spinning NMR. The packing arrangement was studied by 13C dipolar assisted rotational resonance NMR and packing in a staggered arrangement rather than a rectangular arrangement of this peptide with an anti-parallel β-sheet structure was clarified, which is in good agreement with our previous report on the packing arrangement of (Ala)7 with an anti-parallel β-sheet structure.


 Please wait while we load your content...
Please wait while we load your content...