The properties and behavior of α-pinene secondary organic aerosol particles exposed to ammonia under dry conditions
Abstract
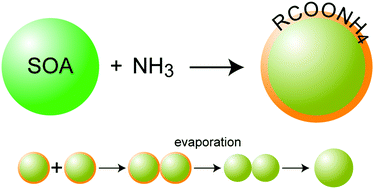
Chemical transformations and aging of secondary organic aerosol (SOA) particles can alter their physical and chemical properties, including particle morphology. Ammonia, one of the common atmospheric reactive constituents, can react with SOA particles, changing their properties and behavior. At low relative humidity, NH3 uptake by α-pinene SOA particles appears to be limited to the particle surface, which suggests that the reacted particles might not be homogeneous and have complex morphology. Here, we present a study aimed at detailed characterization of the effect of ammonia on the composition, density, morphology, shape, and evaporation kinetics of α-pinene SOA particles. We find that a small amount of NH3 diffuses and reacts throughout the particle bulk, while most of the ammoniated products result from the reaction of NH3 with carboxylic acids on the particle surface, leading to a slight increase in particle size. We show that the reaction products form a solid semi-volatile coating that is a few nanometers thick. This solid coating prevents coagulating particles from coalescing for over two days. However, when the gas phase is diluted this semi-volatile coating evaporates in minutes, which is ensued by rapid coalescence. The ammoniated products in the particle bulk affect particles’ evaporation kinetics, more so for the smaller particles that contain a higher fraction of ammoniated products.


 Please wait while we load your content...
Please wait while we load your content...