The effect of the trans axial ligand of cobalt corroles on water oxidation activity in neutral aqueous solutions†
Abstract
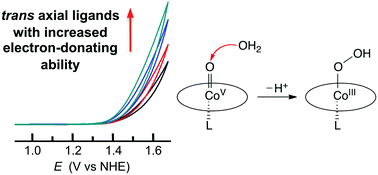
A series of cobalt complexes of 5,10,15-tris(pentafluorophenyl)-corrole [Co(tpfc)] (1) with various axial ligands were synthesized and examined as single-site catalysts for water oxidation. The used axial ligands include 4-cyanopyridine (py-CN), pyridine (py), 4-(dimethylamino)pyridine (py-NMe2), 4-methoxypyridine (py-OMe), 1-methylimidazole (im-Me), and thiophenolate (thi). Complexes 1–py and 1–py-OMe were structurally characterized. The Co ion in both structures has an almost identical distorted octahedral coordination environment with the four N atoms of tpfc defining the equatorial plane and the two molecules of pyridine (for 1–py) or 4-methoxypyridine (for 1–py-OMe) occupying the axial positions. Electrochemical studies of these Co corroles in acetonitrile showed that they all display two oxidation events and the oxidation waves shift to the cathodic direction with electron-donating axial ligands, a trend that is consistent with increased electron densities on Co ions. All these Co corroles were found to be active for electrocatalytic water oxidation: by using catalyst-coated fluorine-doped tin oxide (FTO) working electrodes, cyclic voltammograms displayed pronounced catalytic waves for water oxidation in 0.1 M pH 7.0 phosphate buffer solutions. The onset overpotentials are in the range of 510 to 580 mV, depending on the electron-donating ability of the trans axial ligands. These results demonstrate that the catalytic activities of Co corroles for water oxidation are considerably affected by the trans axial ligands on Co centers and provide valuable insights into the design of new catalysts for water oxidation.


 Please wait while we load your content...
Please wait while we load your content...