Spectral pointillism of enhanced Raman scattering for accessing structural and conformational information on single protein
Abstract
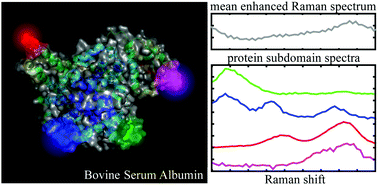
In this contribution, we provide new insights on the temporal fluctuations of surface enhanced Raman spectra (SERS) of large single molecules such as proteins. Because they can only fit partly into small active volume, SERS analysis is referred to spectral pointillism where only protein subdomains are shined and the whole protein landscape is built from the dynamics of successive individual spectra. By applying our approach on bovine serum albumin, we show that single protein subdomains are mostly comprised of three distinct amino acids. Surface amino acids such as lysine are preferentially detected in the open form of the protein. The investigation of the tryptophan Fermi doublet in the single protein regime is highly instructive on the protein conformation. We finally demonstrate that spectral pointillism enables to correlate individual amino acids with structural information.

 Please wait while we load your content...
Please wait while we load your content...