Highly thermally stable heterogeneous catalysts: study of 0D and 3D porphyrinic MOFs†
Abstract
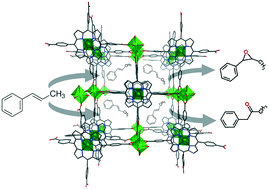
Heterogeneous catalysts are greatly preferred for green chemistry in many industrial processes and, in the past decade, promising results have been achieved in order to improve the catalytic activity of metal organic frameworks (MOFs). Accordingly, porphyrins make the design of new coordination polymers with better properties possible, taking into account the important functions they develop in nature. In this sense, porphyrin-based MOFs are becoming very relevant in heterogeneous catalysis. Thus, the aim of this work was to obtain metalloporphyrinic MOFs exhibiting catalytic activity. Studying the effect of dimensionality on the MOF properties (including thermal stability and catalytic activity), in this work, we study two catalysts with different dimensionalities, 3D [Ni5(H2TCPP)2O(H2O)4]·nS (1) and 0D [Cu(H4TCPP)]·6DMF (2) (where H6TCPP is meso-tetra(4-carboxyphenyl)porphyrin, DMF is N,N-dimethylformamide and S is the solvent). The structural features of both compounds, combined with their high thermal stability and accessible networks, are responsible for their excellent behaviour as heterogeneous catalysts. It is worth mentioning that a significant reduction in reaction time compared to other reported catalysts has been observed. The recyclability of one of the herein studied porphyrin-based MOFs is outstanding. Further structural and thermal characterization has been carried out by means of single crystal X-ray diffraction, IR spectroscopy, thermogravimetry (TG), powder X-ray diffraction (XRD) and transmission electron microscopy (TEM).


 Please wait while we load your content...
Please wait while we load your content...