Crystallization kinetics mechanism investigation of sol–gel-derived NaYF4:(Yb,Er) up-converting phosphors
Abstract
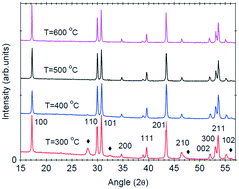
The crystallization mechanism of sol–gel-derived NaYF4:(Yb,Er) up-converting phosphors has been studied by differential scanning calorimetry analysis using both model-free and model fitting approaches. Structural and optical data have shown that the hexagonal NaYF4:(Yb,Er) phase crystallization process occurs at around 315 °C as a result of the thermal decomposition of the metal trifluoroacetates. As the annealing temperature increases, sphere-like microcrystals of about 1–2 μm size (at 300 °C) break up into smaller ones (400–500 nm size) and finally collapse at higher temperatures (600 °C); the up-conversion luminescence signal intensity increases due to the crystallinity improvement and dehydration process. The crystallization process can be described as an autocatalytic-type reaction where the accompanying cubic NaYF4 phase played a catalytic role by reducing the energy barrier against the crystallization of the hexagonal NaYF4 phase, causing its fast self-accelerated crystallization. The energy resulting from the disintegration process of the initial NaYF4 microcrystals contributed to the growth and agglomeration processes and finally the collapse of the crystalline fragments with increasing temperature.

 Please wait while we load your content...
Please wait while we load your content...