Novel drug–drug cocrystals of carbamazepine with para-aminosalicylic acid: screening, crystal structures and comparative study of carbamazepine cocrystal formation thermodynamics†‡
Abstract
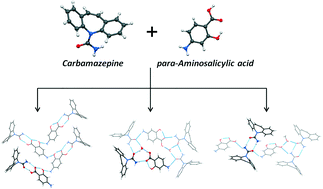
The cocrystal formation of the anticonvulsant drug carbamazepine (CBZ) with para-aminosalicylic acid (PASA, antituberculous drug) has been studied by varying the molar ratios of CBZ to PASA (1 : 1 and 2 : 1) and using liquid-assisted grinding (LAG), slurrying and solution crystallization methods. Three novel cocrystal forms of CBZ and PASA have been synthesized: [CBZ + PASA] (1 : 1), [CBZ + PASA + H2O] (2 : 1 : 1) and [CBZ + PASA + MeOH] (2 : 1 : 1), and their crystal structures have been described. Conformational analysis of the CBZ molecule in its polymorphic forms, its cocrystals and solvates has been conducted. Calculations of intermolecular interaction energies using the PIXEL approach have been carried out for CBZ cocrystals with 1 : 1 stoichiometry. The melting and desolvation processes of the [CBZ + PASA] (1 : 1), [CBZ + PASA + H2O] (2 : 1 : 1) and [CBZ + PASA + MeOH] (2 : 1 : 1) cocrystals have been studied. The temperature dependence of the CBZ (form III) saturation vapor pressure has been studied, and the sublimation thermodynamic functions have been calculated. Based on the sublimation thermodynamics database of molecular crystals, the standard sublimation thermodynamic functions of PASA have been evaluated. The thermodynamic functions of cocrystal formation based on CBZ have been calculated and analyzed. The dissolution process of the [CBZ + PASA] cocrystal (1 : 1) in water (pH 7.4) has been studied. CBZ cocrystallization with PASA has been shown to lead to a dramatic decrease of the CBZ rate of conversion from the anhydrous to the hydrate form, and as a consequence, to solubility improvement by approximately 1.5 times.


 Please wait while we load your content...
Please wait while we load your content...