Towards improved quantitative analysis using surface-enhanced Raman scattering incorporating internal isotope labelling†
Abstract
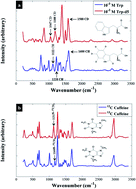
Raman spectroscopy has attracted considerable interest during the past two decades as a vibrational technique used for the molecular characterisation of different molecules. Whilst the Raman effect is known to be generally weak, it is also known that this can be greatly improved using surface-enhanced Raman scattering (SERS). Indeed, in recent years, the power of SERS for rapid identification and quantification of target analytes in a wide range of applications has been repeatedly demonstrated in multiple studies. Moreover, the application of SERS in combination with an isotopically labelled compound (ILC), as an internal standard, has also very recently shown promising results for quantitative SERS measurements, by improving both its accuracy and precision. This is due to the 12C and 13C or 1H and 2H (D) having similar physicochemical properties. The use of these internal standards results in the reduction of any influences due to the number of nanoparticles within the analysis zone and fluctuations in laser fluence. Thus, in this study we have employed SERS for quantitative detection of tryptophan (Trp) and caffeine. These have been chosen because Trp is readily available as the deuterated form and caffeine is available in both 12C and 13C. Quantum chemical calculations based on density functional theory (DFT) have been utilized to determine the vibrational characteristics of the target analytes. For SERS analysis incorporating isotopologues of tryptophan three independent experiments were conducted with three different batches of nanoparticles over a 12 month period; our results show that the use of this internal standard improves quantification of this target molecule. In particular for the independent test sets (i.e., samples not used in quantitative partial least squares regression (PLSR) model construction) we observed improvements in the linearity for test set predictions, as well as lower errors in test set predictions, when isotope internal standards were used during SERS for both deuterated tryptophan as well as 13C caffeine. This work is an extension of and a natural progression from our earlier studies. By exploring additional analytes of interest, allowing for the assessment of the different types of stable isotopes as internal standards, and demonstrating the transfer/robustness of isotopologues for use with SERS, we believe this approach could be readily extended to other biologically-relevant compounds.


 Please wait while we load your content...
Please wait while we load your content...