Microchip-based 3D-cell culture using polymer nanofibers generated by solution blow spinning†
Abstract
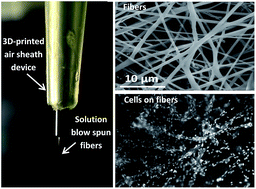
Polymer nano/micro fibers have found many applications including 3D cell culture and the creation of wound dressings. The fibers can be produced by a variety of techniques that include electrospinning, the primary disadvantage of which include the requirement for a high voltage supply (which may cause issues such as polymer denaturation) and lack of portability. More recently, solution blow spinning, where a high velocity sheath gas is used instead of high voltage, has been used to generate polymer fibers. In this work, we used blow spinning to create nano/microfibers for microchip-based 3D cell culture. First, we thoroughly investigated fiber generation from a 3D printed gas sheath device using two polymers that are amenable to cell culture (polycaprolactone, PCL and polystyrene, PS) as well as the parameters that can affect PCL and PS fiber quality. Using the 3D printed sheath device, it was found that the pressure of the sheath N2 and the concentration of polymer solutions determine if fibers can be produced as well as the resulting fiber morphology. In addition, we showed how these fibers can be used for 3D cell culture by directly depositing PCL fibers in Petri dishes and well plates. It is shown the fibers have good compatibility with RAW 264.7 macrophages and the PCL fiber scaffold can be as thick as 178 ± 14 μm. PCL fibers created from solution blow spinning (with the 3D printed sheath device) were then integrated with a microfluidic device for the first time to fabricate a 3D cell culture scaffold with a flow component. After culturing and stimulating macrophages on the fluidic device, it was found that the integrated 3D fibrous scaffold is a better mimic of the extracellular matrix (as opposed to a flat, 2D substrate), with enhanced nitrite accumulation (product of nitric oxide release) from macrophages stimulated with lipopolysaccharide. PS fibers were also made and integrated in a microfluidic device for 3D culture of endothelial cells, which stayed viable for at least 72 hours (48 hours under the flowing conditions). This approach will be useful for future studies involving more realistic microchip-based culture models for studying cell-to-cell communication.

 Please wait while we load your content...
Please wait while we load your content...