A dual responsive “turn-on” fluorophore for orthogonal selective sensing of biological thiols and hydrogen peroxide†
Abstract
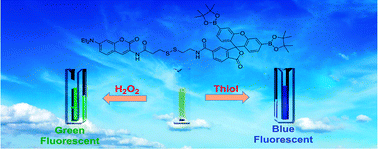
Both thiols and hydrogen peroxide (H2O2) have great correlations with cancer and other diseases, and hence detection probes for sensing these agents may serve as early diagnostic tools. In this article, we report the development of a dual responsive probe that has the ability to generate two different responses upon reacting with thiols and H2O2 in a highly selective manner. The probe (FLB2SSCou) consists of a coumarin unit and a diboron xanthene spiro isobenzofuran group bridged by a disulfide bond. The detection experiments show that the probe could selectively respond to thiols and H2O2 when screening a substrate library containing 20 amino acids, homocysteine, glutathione, dithiothreitol and H2O2. The initial off state of the probe was a result of photo-induced electron transfer (PET) from the coumarin group to the non-fluorescing diboron xanthene spiro isobenzofuran group bridged by a disulfide bond. Reductive cleavage of the disulfide bond leads to the termination of this PET process, thus switching on the fluorescence of the probe. On the other hand, the oxidation of the diboron group by H2O2 converts the non-fluorescing group into a highly fluorescing fluorescein group. Time-dependent density functional theory calculations were then performed to explain the PET process, and the obtained results indicate that the PET process occurs from the second excited state (S2) into the first excited state (S1). Finally, imaging and detection experiments of the probe on HeLa cancer cells were conducted by means of the fluorescence microscopy and flow cytometry technique. It was observed that the fluorescence of the FLB2SSCou probe could be switched on by endogenous thiols and exogenous H2O2, demonstrating the applicability of this probe in both extracellular and intracellular environments. The present work exhibits the novel development of a dual responsive probe in contrast to commonly reported single responsive fluorescent probes, which may inspire the future design of multiple responsive fluorescent probes.
- This article is part of the themed collection: Shape-Responsive Fluorophores

 Please wait while we load your content...
Please wait while we load your content...