Chain mobility and film softness mediated protein antifouling at the solid–liquid interface
Abstract
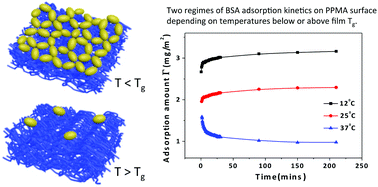
The influences of polymer chain mobility (flexibility) and film softness on protein adsorption at the solid/liquid interface were studied with a series of polymethacrylate (PMA) polymers (PMMA, PEMA, PPMA, PnBMA) which have similar chemical structures but different glass transition temperatures (Tgs). It is found that these PMA films all adsorb protein molecules when the adsorption temperature is lower than their Tgs, and the adsorption amount varies in response to different mechanical properties of PMAs, i.e. stiffness, modulus, deformation and adhesion as measured by scanning probe microscopy. PnBMA, which has the longest side chain and lowest stiffness, adsorbed the lowest amount of protein molecules, although it is the most hydrophobic polymer among these four PMAs. There is a significant reduction of protein adsorption on a polymer film in the rubbery state, even when the adsorption temperature is only 1.5 °C higher than the Tg. Our experiments suggest that the chain mobility and film softness of polymers can influence protein adsorption at the solid–liquid interface, and even overwhelm the hydrophilic effect under certain conditions. Polymers with high chain mobility and softness provide superior protein antifouling properties as a result of the high entropy barrier from film surfaces.

 Please wait while we load your content...
Please wait while we load your content...