Maghemite-human serum albumin hybrid nanoparticles: towards a theranostic system with high MRI r2* relaxivity†
Abstract
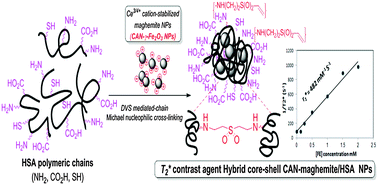
Human Serum Albumin (HSA) is the most abundant plasma protein in human blood, and therefore, it is the material of choice for the development of particulate formulations due to its biodegradable and biocompatible nature. Over the last decade, HSA nanoparticles (NPs) have been prepared mostly using desolvation techniques and evaluated as promising drug carriers. In addition, controlling the particle size has become a primary concern while formulating such nanoparticulate systems. Since many of these HSA-based carrier systems have often demonstrated batch-to-batch fabrication variability, significant efforts have been made to develop and characterize HSA-based NPs featuring a robust and controllable particle size, by using a desolvation/cross-linking-type Divinyl Sulfone (DVS)-mediated nanofabrication method. For this purpose and for global multi-parameter fabrication process optimization, a statistically significant Design of Experiment (DoE, MINITAB® 17 DoE software) methodology has been successfully implemented. It aimed to disclose an optimal set of HSA NP fabrication conditions in order to afford highly reproducible and stable 23.05 ± 5.3 nm-sized DoE-globally optimized core HSA NPs. Due to the use of bifunctional DVS as a cross-linker for the preparation of such DoE-optimized HSA NPs, their surface contains a variety of free functional groups which are available for further second step functional modifications. Moreover, related hybrid organic/inorganic nanosystems consisting of DoE-optimized HSA NPs that encapsulated hydrophilic (NH4)2Ce(IV)(NO3)6 (Ceric Ammonium Nitrate – CAN) modified γ-Fe2O3 NPs (CAN-maghemite or CAN-γ-Fe2O3 NPs), which enable medical imaging using magnetic resonance imaging (MRI), have also been fabricated and characterized. The resulting hybrid magnetic NPs are a quite powerful T2* contrast agent (r2* of 482 mM−1 s−1), which may be used as a powerful dual phase platform for both therapeutic (drug delivery) and diagnostic imaging (MRI) applications.

 Please wait while we load your content...
Please wait while we load your content...