Effect of the protein corona on the colloidal stability and reactivity of LDH-based nanocarriers†
Abstract
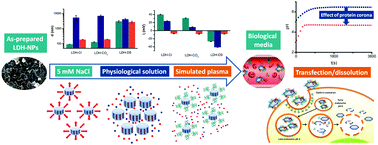
The physicochemical properties of drug nanocarriers such as layered double hydroxide nanoparticles (LDH-NPs) determine their circulation times in biological media and their interaction with the targeted cells. Nevertheless, the components of the biological fluid, and particularly the formation of a protein corona, change the properties of as-prepared nanocarriers. Here, we discuss the effect of the protein corona formation on the colloidal stability and reactivity of LDH-NPs intercalated with chloride (LDH–Cl), carbonate (LDH–CO3) or dodecylsulfate (LDH–DS). These solids present model physicochemical properties (hydrophillic character, surface charge, and exchange capacity) that can be obtained depending on the interaction of drugs with LDH layers. The colloidal stability of LDH-NPs was determined in simulated biological fluids at high ionic strength and/or the presence of albumin (the main protein of human blood plasma), whereas the reactivity was evaluated by dissolution kinetics in acidic media, compatible with the environment of cell internalized nanocarriers. The protein corona increased the colloidal stability of the nanocarriers by steric hindrance at high ionic strength, reverted the positive zeta potential of as-prepared LDH-NPs and protected them from dissolution at low pHs. The properties of the anionic cargo of LDH-NPs strongly affected the protein corona and hence the fate of NPs in biological fluids. Drug nanocarriers with interfacial properties similar to those of LDH–Cl and LDH–CO3 seem to be more promising than LDH–DS in forming a protein corona. Then, LDH–Cl and LDH–CO3 would enable long circulation times due to their size, colloidal stability and low protein damage. Our results indicate that LDH-NPs preserve and even improve their properties as drug nanocarriers after interacting with the biological media, particularly their ability to reach the site of therapeutic action from the injection place.

 Please wait while we load your content...
Please wait while we load your content...