Understanding the contribution of surface roughness and hydrophobic modification of silica nanoparticles to enhanced therapeutic protein delivery†
Abstract
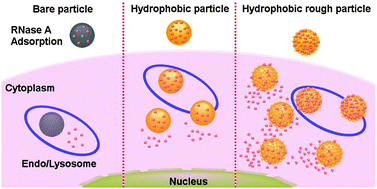
Intracellular protein delivery holds great promise for cancer therapy. In this work, the individual and combined contribution of the surface roughness and hydrophobic modification (octadecyl-group) of silica nanoparticles has been studied in a number of events for cellular delivery of therapeutic proteins, including loading capacity, release behaviour, cellular uptake and endo/lysosomal escape. Both surface roughening and hydrophobic modification enhance the protein adsorption capacity and sustained release, while the contribution from the surface roughness is higher for loading capacity and hydrophobic modification is more effective for sustained protein release. Both structural parameters improve the cellular uptake performance; however the difference in the contribution is cell type-dependent. Only the hydrophobic modification shows a contribution to endo/lysosomal escape, independent of the surface topography. Octadecyl-functionalized rough silica nanoparticles thus show the best performance in therapeutic protein (RNase A) delivery, causing significant cell viability inhibition in different cancer cells among all groups under study.

 Please wait while we load your content...
Please wait while we load your content...