Cobalt nickel phosphide nanoparticles decorated carbon nanotubes as advanced hybrid catalysts for hydrogen evolution†
Abstract
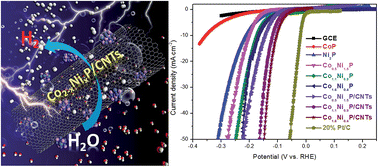
Exploring new hybrid catalysts to replace Pt-based catalysts for the hydrogen evolution reaction (HER) is important for various renewable energy applications. However, the design and synthesis of such catalysts are still challenging. Herein, we focus on the development of a series of hybrid materials consisting of cobalt nickel phosphide nanoparticles (NPs) decorated carbon nanotubes (Co2−xNixP/CNTs) as efficient catalysts for enhanced HER catalytic activity. All the X-ray spectra including X-ray diffraction, X-ray photoelectron spectroscopy and X-ray adsorption spectroscopy demonstrate that the crystalline phase structure, valence and coordination environment of hexagonal Ni2P are changed with increasing Co atoms. Electrochemical measurements show that the Co2−xNixP/CNT hybrids exhibit high activity and stability for the HER in acidic solution. The as-synthesized Co1.6Ni0.4P/CNT hybrid exhibits the highest electrocatalytic activity with low onset overpotential (36.1 mV), a small Tafel slope (46.7 mV dec−1), a much larger exchange current density (1.86 × 10−5 A cm−2), lower HER activation energy (57.3 kJ mol−1), and good stability. Such enhanced catalytic activity originates from the introduction of Co and strong synergistic effects between CNTs and Co2−xNixP. Meanwhile, density functional theory calculations also confirm that the higher HER catalytic activity of the Co2−xNixP/CNTs can be attributed to the splendid migratory aptitude of adsorbed single H atoms and the lower energy barrier for H2 formation after the introduction of Co atoms. The as-synthesized Co2−xNixP/CNT hybrid catalysts have great potential practical application in water splitting.

 Please wait while we load your content...
Please wait while we load your content...