Highly proton conductive, dense polybenzimidazole membranes with low permeability to vanadium and enhanced H2SO4 absorption capability for use in vanadium redox flow batteries†
Abstract
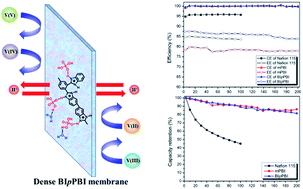
H2SO4-doped dense polybenzimidazole (PBI) membranes based on poly[2,2′-(2-benzimidazole-p-phenylene)-5,5′-bibenzimidazole] (BIpPBI) containing benzimidazole side groups were developed for use in vanadium redox flow batteries (VRFBs). BIpPBI was prepared by the polycondensation of 3,3′-diaminobenzidine with 2-benzimidazole terephthalic acid (BITA) in polyphosphoric acid. Many basic properties of BIpPBI were characterized and compared with conventional PBI (mPBI). The BIpPBI membrane has an entirely amorphous structure because of the presence of additional benzimidazole side groups, and the absorption of both H2SO4 (65 wt%) and water (46 wt%) was significantly improved compared to the mPBI membrane in a 4 M H2SO4 solution. The H2SO4-doped BIpPBI membrane has a low area resistance of 0.17 Ω cm2 and significantly lowered permeability to vanadium ions (3.45 × 10−8 cm2 min−1). Finally, the VRFB assembled with BIpPBI had higher coulomb efficiencies (>99%) and energy efficiencies (78–95%) than Nafion 115 under a wide range of current densities (20–100 mA cm−2). More importantly, the VRFB with BIpPBI exhibited stable cycling performance, running for 200 charge–discharge cycles with 4.0% energy efficiency decay and 81.5% capacity retention. Furthermore, the BIpPBI membrane was stable in a highly oxidizing VO2+ solution, demonstrating its outstanding physicochemical stability. All experimental results indicate that the dense BIpPBI membrane is a promising material for VRFB applications.

 Please wait while we load your content...
Please wait while we load your content...