Film morphology evolution during solvent vapor annealing of highly efficient small molecule donor/acceptor blends†
Abstract
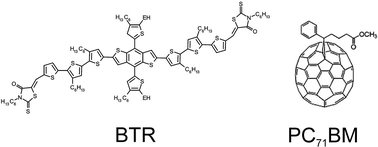
Solution-processable small molecule photovoltaics based on the novel molecular donor, benzodithiophene terthiophene rhodanine (BTR), recently have shown maximum power conversion efficiencies above 8% for active layer thicknesses up to 400 nm, using post process solvent vapor annealing (SVA) with tetrahydrofuran (THF). Here we report an in situ study on the morphology evolution during SVA using the moderate solvent THF and the good solvent chloroform (CF). The combination of real-time grazing incidence X-ray diffraction (GIXD) and grazing incidence small angle X-ray scattering (GISAXS) allows us to draw a complete picture of the evolution of crystallinity and phase purity during post process annealing. We find that the relative crystallinity compared to the as-cast films is only modestly affected by SVA and solvent choice. However, both the phase purity and the characteristic domain sizes within the film vary significantly and are controlled by the solvent quality as well as exposure time. Using THF, films with high phase purity and desirable characteristic length scales of about 30 nm can be achieved, while the use of CF rapidly leads to excessive film coarsening and less preferable domain sizes on the order of 60 nm, too large for optimized charge separation.

 Please wait while we load your content...
Please wait while we load your content...