First-principles and experimental study of nitrogen/sulfur co-doped carbon nanosheets as anodes for rechargeable sodium ion batteries†
Abstract
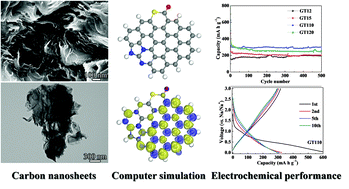
Heteroatom doped carbon materials have recently demonstrated an outstanding sodium storage ability and are being considered as the most promising candidate as anodes for sodium ion batteries. However, there is limited understanding of the relationship between structural and electronic properties and electrochemical storage capacity. First-principles calculations on a doped graphene cluster propose that N, S co-doping can promote the electronegativity, adsorption capacity of Na atoms and diffusion of Na+ ions on graphene sheets, especially for the sample consisting of more pyridinic-N, while excessive O atoms may alleviate these. All these features render N, S co-doped carbon as a superior anode for sodium ion batteries. Therefore, the N, S co-doped carbon nanosheets are fabricated via a simple thermal treatment method using gelatin as the carbon source and thiourea as the N and S precursor. The optimized product (mgelatin : mthiourea = 1 : 10) results in a superb cycling capacity of 300 mA h g−1 after 500 cycles, with a coulombic efficiency of ∼100%. This study provides a facile and reliable route to prepare co-doped carbon with enhanced sodium storage properties.


 Please wait while we load your content...
Please wait while we load your content...