Rational design of cobalt–chromium layered double hydroxide as a highly efficient electrocatalyst for water oxidation†
Abstract
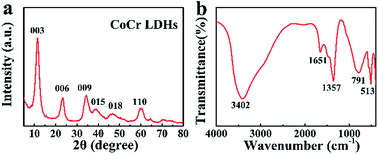
The design of a high performance, stable and cost-effective electrocatalyst for oxygen evolution is crucial for H2 production from electrochemical water splitting. Here, as a dual-functional-site catalyst, cobalt–chromium layered double hydroxide (CoCr LDH) nanosheets are designed and synthesized, where Co2+ is the catalytically active site and Cr3+ is the charge transfer site. OER investigation of CoCr LDH is conducted for the first time. The CoCr LDH nanosheets have a high specific surface area of 151.78 m2 g−1 and exhibit outstanding OER activities, among the best of Co-based candidates. Accordingly, our catalyst affords a low onset potential of 1.47 V (vs. reversible hydrogen electrode, RHE) and a stable current density of 22.8 mA cm−2 at 1.61 V (vs. RHE) for 12 h. The Tafel slope of CoCr LDH is 81.0 mV dec−1, smaller than that of state-of-the-art RuO2 (90.1 mV dec−1). Therefore, the CoCr LDH nanosheets are promising OER catalysts.

 Please wait while we load your content...
Please wait while we load your content...