Efficient ceria nanostructures for enhanced solar fuel production via high-temperature thermochemical redox cycles†
Abstract
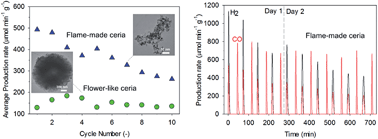
Syngas synthesis by solar energy-driven two-step thermochemical redox cycles is a promising approach for large-scale industrial production of renewable fuels. A key challenge is developing durable materials capable of providing and sustaining high redox kinetics under harsh environmental conditions required for efficient operation. Here, we demonstrate that nanostructured ceria with a high surface area and porosity can significantly enhance the initial and long-term syngas production performance. Three types of ceria morphologies were synthesised and comparatively investigated against commercial powders in two-step thermochemical redox cycles, namely nanostructured flame-made and flower-like agglomerates and sol–gel sub-micro particles. Their syngas production performance was assessed in terms of redox kinetics, conversion stoichiometry and structural stability. The flame-made ceria nano-powders had up to 191%, 167% and 99% higher initial average production rates than the flower-like, commercial and sol–gel ceria powders, respectively. This resulted in the highest H2 (480 μmol min−1 g−1) and CO (230 and 340 μmol min−1 g−1) production rates and redox capacity (Δδ = 0.25) so far reported for ceria. Notably, the grain morphology played a key role in the long-term performance and while the redox kinetics of the flower-like ceria rapidly decreased below that of the commercial powders, the flame-made agglomerates maintained up to 57% higher average production rate until the last cycle. These findings show that the thermochemical stabilisation of nano-scale structural features, observed in the flame-made agglomerates, is key to engineering efficient materials for enhanced thermochemical solar fuel production.

 Please wait while we load your content...
Please wait while we load your content...