Effects of moisture on (La, A)MnO3 (A = Ca, Sr, and Ba) solid oxide fuel cell cathodes: a first-principles and experimental study
Abstract
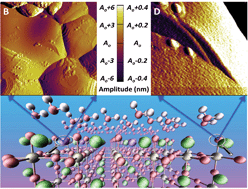
One of the major challenges in developing clean, environmentally friendly energy technologies such as solid oxide fuel cells (SOFCs) is performance degradation at higher temperatures. Solid oxide fuel cell (SOFC) cathode degradation in the presence of moisture is one of the major concerns. Combining computational and experimental studies provides a comprehensive picture of the interaction between moisture and lanthanum manganite based SOFC cathodes. To better understand the surface chemistry, (La, A)MnO3 (A = Ca, Sr and Ba) (001) surfaces are explored using first-principles calculations. This computational study suggests that dissociative adsorption of water molecules is favored on (La, A)O-terminated (001) surfaces. The covalently unsaturated surface terminal O atom (via strong H-bond) attracts one of the H atoms of the water molecule, with a subsequent breakup of the water molecule into H+ and OH− groups. The surface should also be significantly enriched with A-site dopants under all realistic conditions, with all the three dopants driven to segregate to the surface over a wide range of T–pH2O conditions. Atomic force microscopy reveals just such a segregation of dopants to the surface of doped LaMnO3, enhanced in the presence of moisture. It is hypothesized that the interplay of the resulting oxygen vacancy defects and moisture from the operating environment further drives cationic surface segregation, ultimately degrading catalytic activity. In addition to providing insights into the surface chemistry, this combined experimental and computational investigation opens pathways for designing new materials with enhanced catalytic functionality.

 Please wait while we load your content...
Please wait while we load your content...