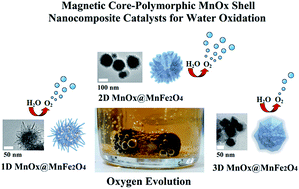
Water oxidation catalysis by using nano-manganese ferrite supported 1D-(tunnelled), 2D-(layered) and 3D-(spinel) manganese oxides†
Abstract
We report a new series of core–shell composite catalysts obtained by supporting 1D-(tunnelled), 2D-(layered) and 3D-(spinel) manganese oxides on nano-manganese ferrite. All other factors being equal, magnetic 2D-MnOx structures carried out the chemical water oxidation reaction with Ce4+ at a higher efficiency than their 1D-, 3D- and non-magnetic counterparts. Ca-birnessite, when supported on a ferrite core, produced the highest amount of O2 [3900 mmol (O2) per mol (Mn) per h] normalized per mol of manganese in the catalysts. The composition and morphology parameters of the magnetic core–shell structures appeared to be important in determining the observed activity while no direct correlation was noted with the surface area.

 Please wait while we load your content...
Please wait while we load your content...