Efficient fullerene-free organic solar cells based on fused-ring oligomer molecules†
Abstract
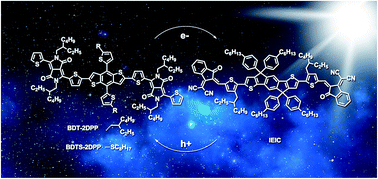
We design and synthesize monodisperse fused-ring oligomer molecules benzo[1,2-b:4,5-b′]dithiophene (BDT) flanked with electron-withdrawing diketopyrrolopyrrole (DPP). A tiny change in the side chain induces significant variation in crystallinity, phase separation, charge transport and photovoltaic properties of the semiconductors. BDTS-2DPP with linear alkylthio substituents exhibits a much higher hole mobility of 1.1 × 10−2 cm2 V−1 s−1 than the branched alkyl substituted BDT-2DPP (3.0 × 10−3 cm2 V−1 s−1). The LUMO (−3.49 eV) and HOMO (−5.28 eV) energy levels of BDTS-2DPP are lower than those of BDT-2DPP (−3.46 eV and −5.23 eV) due to the π-acceptor capability of the sulfur atom. Fullerene-free organic solar cells using BDTS-2DPP as a donor and monodisperse fused-ring oligomer molecule IEIC as an acceptor exhibit higher open-circuit voltage, short-circuit current density, fill factor and power conversion efficiency (PCE, 5.29%) than the BDT-2DPP control devices (PCE = 4.00%) with the conventional structure. The inverted devices based on BDTS-2DPP:IEIC show an improved PCE of 6.03% relative to the conventional devices. Atomic force microscopy, grazing incident wide-angle X-ray diffraction and resonant soft X-ray scattering are used to deeply investigate the molecular packing, phase separation and surface aggregation of the blended films and to understand the effect of molecular side chains. We find that the linear alkylthio substitution in BDTS-2DPP improves the crystallinity and unexpectedly reserves small phase separation domains in the blend.

 Please wait while we load your content...
Please wait while we load your content...