Oxygen evolution reaction electrocatalysis on SrIrO3 grown using molecular beam epitaxy†
Abstract
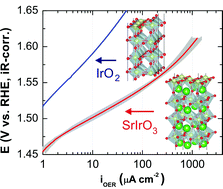
Electrochemical generation of oxygen via the oxygen evolution reaction (OER) is a key enabling step for many air-breathing electrochemical energy storage devices. IrO2 (Ir4+: 5d5) ranks among the most active known OER catalysts. However, it is unclear how the environment of the Ir4+ oxygen-coordination octahedra affects the OER electrocatalysis. Herein, we present the OER kinetics on a single-crystal, epitaxial SrIrO3(100)p perovskite oxide synthesized using molecular-beam epitaxy on a DyScO3(110) substrate. We find that by switching the host structure of the Ir4+ oxygen-coordination octahedra from corner- and edge-sharing rutile (IrO2) to purely corner-sharing perovskite (SrIrO3), the OER activity increases by more than an order of magnitude. We explain our finding with the correlated, semimetal electronic structure of SrIrO3; our density functional theory calculations reveal that the adsorption energetics on SrIrO3 depends sensitively on the electron–electron interaction, whereas for IrO2, it depends rather weakly. This finding suggests the importance of correlations on the OER and the design of future transition metal oxide electrocatalysts.
- This article is part of the themed collection: Emerging Investigators 2016: Novel design strategies for new functional materials

 Please wait while we load your content...
Please wait while we load your content...