Colloidally-synthesized cobalt molybdenum nanoparticles as active and stable electrocatalysts for the hydrogen evolution reaction under alkaline conditions†
Abstract
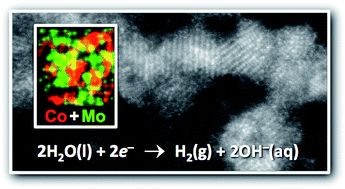
The efficient catalysis of water electrolysis using Earth-abundant materials is considered to underpin the large-scale implementation of several clean energy technologies. Here, we report the synthesis of molybdenum rich Co–Mo nanoparticles, which function as highly active and stable electrocatalysts for the hydrogen evolution reaction (HER) in alkaline aqueous solutions. The Co–Mo nanoparticles have an average diameter of approx. 3 nm, a Co : Mo ratio of approx. 1 : 9, and are poorly crystalline. The Co–Mo nanoparticles deposited onto Ti foil substrates at a loading of approx. 1 mg cm−2 required an overpotential of only −75 mV to produce an operationally relevant current density of −10 mA cm−2, placing them among the most highly active Earth-abundant HER catalysts in alkaline media.
- This article is part of the themed collection: Water splitting and photocatalysis

 Please wait while we load your content...
Please wait while we load your content...