Swelling of phospholipid membranes by divalent metal ions depends on the location of the ions in the bilayers†
Abstract
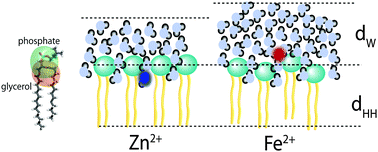
The Hofmeister series illustrates how salts produce a wide range of effects in biological systems, which are not exclusively explained by ion charge. In lipid membranes, charged ions have been shown to bind to lipids and either hydrate or dehydrate lipid head groups, and also to swell the water layer in multi-lamellar systems. Typically, Hofmeister phenomena are explained by the interaction of the ions with water, as well as with biological interfaces, such as proteins or membranes. We studied the effect of the divalent cations Mg2+, Ca2+, Fe2+, and Zn2+ on oriented, stacked, phospholipid bilayers made of dimyristoylphosphatidylcholine (DMPC). Using high-resolution X-ray diffraction, we observed that the cations lead to a swelling of the water layer between the bilayers, without causing significant changes to the bilayer structure. The cations swelled the bilayers in different amounts, in the order Fe2+ > Mg2+ > Ca2+ > Zn2+. By decomposing the total bilayer electron density into different molecular groups, Zn2+ and Ca2+ were found to interact with the glycerol groups of the lipid molecules and cause minor swelling of the bilayers. Mg2+ and Fe2+ were found to position near the phosphate groups and cause a strong increase in the number of hydration water molecules. Our results present a molecular mechanism-of-action for the Hofmeister series in phospholipid membranes.
- This article is part of the themed collection: Open access articles from Soft Matter

 Please wait while we load your content...
Please wait while we load your content...