AFM mapping of the elastic properties of brain tissue reveals kPa μm−1 gradients of rigidity†
Abstract
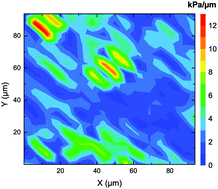
It is now well established that the mechanical environment of the cells in tissues deeply impacts cellular fate, including life cycle, differentiation and tumor progression. Designs of biomaterials already include the control of mechanical parameters, and in general, their main focus is to control the rheological properties of the biomaterials at a macroscopic scale. However, recent studies have demonstrated that cells can stress their environment below the micron scale, and therefore could possibly respond to the rheological properties of their environment at this micron scale. In this context, probing the mechanical properties of physiological cellular environments at subcellular scales is becoming critical. To this aim, we performed in vitro indentation measurements using AFM on sliced human pituitary gland tissues. A robust methodology was implemented using elasto-adhesive models, which shows that accounting for the adhesion of the probe on the tissue is critical for the reliability of the measurement. In addition to quantifying for the first time the rigidity of normal pituitary gland tissue, with a geometric mean of 9.5 kPa, our measurements demonstrated that the mechanical properties of this tissue are far from uniform at subcellular scales. Gradients of rigidity as large as 12 kPa μm−1 were observed. This observation suggests that physiological rigidity can be highly non-uniform at the micron-scale.

 Please wait while we load your content...
Please wait while we load your content...