Polysulfide-1-oxides react with peroxyl radicals as quickly as hindered phenolic antioxidants and do so by a surprising concerted homolytic substitution†
Abstract
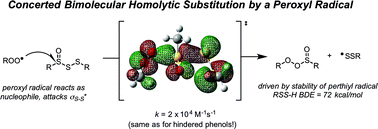
Polysulfides are important additives to a wide variety of industrial and consumer products and figure prominently in the chemistry and biology of garlic and related medicinal plants. Although their antioxidant activity in biological contexts has received only recent attention, they have long been ascribed ‘secondary antioxidant’ activity in the chemical industry, where they are believed to react with the hydroperoxide products of autoxidation to slow the auto-initiation of new autoxidative chain reactions. Herein we demonstrate that the initial products of trisulfide oxidation, trisulfide-1-oxides, are surprisingly reactive ‘primary antioxidants’, which slow autoxidation by trapping chain-carrying peroxyl radicals. In fact, they do so with rate constants (k = 1–2 × 104 M−1 s−1 at 37 °C) that are indistinguishable from those of the most common primary antioxidants, i.e. hindered phenols, such as BHT. Experimental and computational studies demonstrate that the reaction occurs by a concerted bimolecular homolytic substitution (SH2), liberating a perthiyl radical – which is ca. 16 kcal mol−1 more stable than a peroxyl radical. Interestingly, the (electrophilic) peroxyl radical nominally reacts as a nucleophile – attacking the 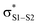 of the trisulfide-1-oxide – a role hitherto suspected only for its reactions at metal atoms. The analogous reactions of trisulfides are readily reversible and not kinetically competent to inhibit hydrocarbon autoxidation, consistent with the longstanding view that organosulfur compounds must be oxidized to afford significant antioxidant activity. The reactivity of trisulfides and their oxides are contrasted with what is known of their shorter cousins and predictions are made and tested with regards to the reactivity of higher polysulfides and their 1-oxides – the insights from which may be exploited in the design of future antioxidants.
of the trisulfide-1-oxide – a role hitherto suspected only for its reactions at metal atoms. The analogous reactions of trisulfides are readily reversible and not kinetically competent to inhibit hydrocarbon autoxidation, consistent with the longstanding view that organosulfur compounds must be oxidized to afford significant antioxidant activity. The reactivity of trisulfides and their oxides are contrasted with what is known of their shorter cousins and predictions are made and tested with regards to the reactivity of higher polysulfides and their 1-oxides – the insights from which may be exploited in the design of future antioxidants.

 Please wait while we load your content...
Please wait while we load your content...